According to Nova One Advisor, the global allogeneic cell therapy market size was valued at USD 528.65 million in 2025 and is poised to grow from USD 673.66 million in 2026 to USD 5968.77 million by 2035, at a CAGR of 27.43% during the forecast period (2026-2035).
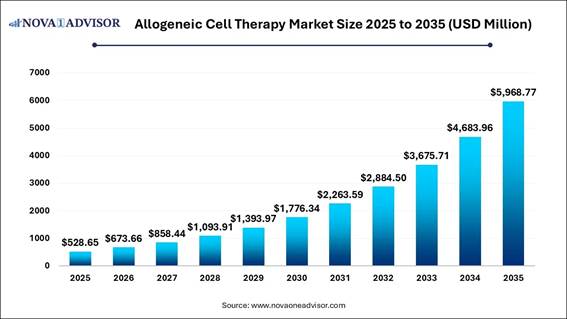
From experimental laboratory findings to readily available, off-the-shelf patient care, the allogeneic cell therapy market is experiencing a significant transformation, moving from personalized autologous methods to scalable, off-the-shelf solutions that are revolutionizing regenerative medicine. This shift is driven by the demand for accessible treatments, utilizing healthy donor sources, CRISPR gene editing, and automated GMP-compliant bioreactors to create universal, immune-evasive cells. As a result, production time has been reduced from months to weeks for treating aggressive cancers and degenerative diseases.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/7933
Key Takeaways
· North America dominated with the largest market share in 2025.
· Europe is anticipated to have the fastest growth with a notable CAGR during the forecast period.
· By therapy type, the stem cell therapy segment contributed the highest market share in 2025.
· By therapy type, the non-stem cell therapy segment is growing at a strong CAGR between 2026 and 2035.
· By therapeutic area, the hematological disorders segment accounted for the largest market share in 2025.
· By therapeutic area, the dermatological disorders segment is projected to grow at a solid CAGR between 2026 and 2035.
Market Overview: The Next Generation of Donor-Derived Cell Therapies
Allogeneic cell therapy involves the use of cells, typically stem cells or immune cells, sourced from healthy donors rather than the patient. This approach allows for off-the-shelf availability and more cost-effective manufacturing compared to autologous therapies. The market is rapidly growing due to advancements in gene editing that help overcome immune rejection, alongside an increasing demand for accessible solutions to cancer and chronic diseases. This offers a scalable and cost-effective future for life-saving medical care.
The Synergy of iPSCs and CRISPR: Major Potential
The stem cell therapy market is increasingly focusing on induced pluripotent stem cells (iPSCs) as a more ethical alternative to embryonic cells. When combined with CRISPR/Cas9 gene editing, this approach allows for precise correction of genetic mutations in regenerative medicine. This synergy facilitates the development of scalable, off-the-shelf allogeneic therapies, promoting market growth by replacing expensive, patient-specific treatments with standardized, mass-produced solutions.
The High Cost of Scaling: Major Limitation
Despite promising clinical results, the stem cell therapy market faces challenges due to high manufacturing costs and scalability issues. The complex processes required for good manufacturing practice (GMP) compliance are labor-intensive and come with prohibitive costs, limiting patient access and presenting significant technical hurdles in maintaining stability and safety during large-scale production.
Immediate Delivery is Available | Get Full Report Access@ https://www.novaoneadvisor.com/report/checkout/7933
Report Scope of Allogeneic Cell Therapy Market
|
Report Coverage |
Details |
|
Market Size in 2026 |
USD 673.66 Million |
|
Market Size by 2035 |
USD 5968.77 Million |
|
Growth Rate From 2026 to 2035 |
CAGR of 27.43% |
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
Segments Covered |
Therapy Type, Therapeutic Area, Region |
|
Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
|
Regional scope |
North America, Europe, Asia Pacific, Latin America, MEA |
|
Key Companies Profiled |
SSM Cardinal Glennon Children's Medical Center; Cleveland Cord Blood Center; Duke University School of Medicine; New York Blood Center; Clinimmune Labs; University of Colorado Cord Blood Bank; MD Anderson Cord Blood Bank; LifeSouth Community Blood Centers, Inc.; Bloodworks Northwest; JCR Pharmaceuticals Co., Ltd.; Sumitomo Pharma Co., Ltd.; Atara Biotherapeutics; Mallinckrodt Pharmaceuticals; Tego Science Inc; Takeda Pharmaceutical Company Limited; STEMPEUTICS RESEARCH PVT LTD; Biosolution Co., Ltd.; MEDIPOST Co., Ltd. |
You can place an order or ask any questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344
Allogeneic Cell Therapy Market: Regional Analysis
In 2025, North America is expected to dominate the market, driven by early technology adoption, significant investment, and advanced regulatory frameworks. The region boasts a robust biotechnology infrastructure, numerous GMP-compliant manufacturing facilities, and a concentration of leading research institutions and pharmaceutical companies. The proactive approach of the U.S. FDA, including the Regenerative Medicine Advanced Therapy (RMAT) designation and expedited approval pathways, supports clinical translation and commercialization.
The U.S. leads the allogeneic cell therapy market, fueled by major biotechnology hubs and substantial investments in CAR-T and NK cell therapies. Innovators like Allogene Therapeutics, Fate Therapeutics, and Atara are at the forefront of developing off-the-shelf, scalable immunotherapy solutions for cancers and other diseases.
· In November 2025, the FDA's RMAT Designation will provide a fast-track pathway for groundbreaking regenerative medicine therapies, expediting the market entry of innovative cell and gene therapies that address serious, unmet medical needs while fostering advancements in manufacturing.
Canada is emerging as a global leader in allogeneic stem cell therapies, thanks to a strong healthcare ecosystem and a focus on regenerative medicine that supports significant market growth. The country is effectively bridging the gap between innovative research and commercialization through specialized organizations committed to achieving commercial-grade GMP manufacturing authorization.
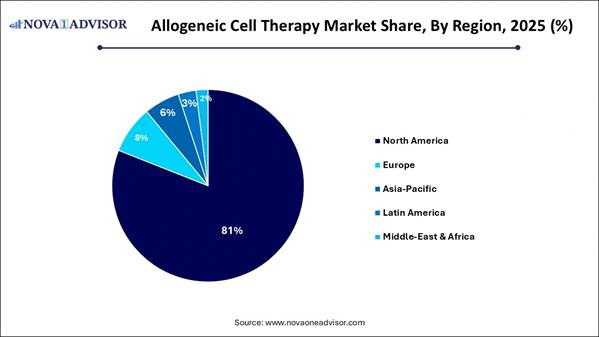
Europe is predicted to experience the fastest growth during the forecast period, primarily due to a major shift toward allogeneic therapies that offer improved scalability, lower costs, and quicker treatment times compared to autologous options. Innovations in CRISPR and TALEN technologies are enhancing safety and effectiveness by reducing issues such as immune rejection and graft-versus-host disease (GvHD). The European Medicines Agency and local regulators are also providing clearer guidelines and faster approval processes for advanced therapy medicinal products.
Germany is solidifying its leadership in the allogeneic cell therapy market by merging top-tier regenerative medicine research with a robust infrastructure, supported by funding from the BMBF and the German Stem Cell Network. The National Strategy for Gene and Cell Therapies, along with regional hubs in North Rhine-Westphalia and Saxony, is accelerating translational research and biomanufacturing in the country.
France is positioning itself as a leading European hub for allogeneic CAR-T development, fueled by significant public investment through the PEPR Biotherapies and Biomanufacturing program. Key advancements in this ecosystem include Allogenica’s grant-funded universal CAR-T research and the CellAction platform at Institut Curie, while startups like TreeFrog Therapeutics are accelerating production scaling.
Allogeneic Cell Therapy Market: Segmental Analysis
By Therapy Type Analysis
The stem cell therapy segment dominated the market in 2025, primarily due to its efficacy in treating chronic diseases like leukemia and other blood cancers, as well as its off-the-shelf availability. There is a high demand for treatments for cancers, blood disorders, and musculoskeletal issues, such as osteoarthritis, which drives the adoption of stem cell therapies. The leading segment is adipose tissue-derived stem cells, closely followed by bone marrow and cord-derived MSCs, due to their therapeutic versatility. The increasing use of automated bioreactors for manufacturing, combined with advancements in gene editing, has resulted in higher yields and improved safety profiles.
The non-stem cell therapy segment is expected to experience the fastest growth during the forecast period. This is mainly driven by the rapid expansion of off-the-shelf CAR-T clinical studies and their superior success rates in targeting solid tumors and autoimmune diseases. Additionally, there has been a significant rise in clinical trials evaluating gene-edited T-cells and Natural Killer cells to treat hematological malignancies. Advancements in CRISPR-Cas9 and TALEN technologies enable precise genetic modifications that create universal cells, reducing the risk of graft-versus-host disease and enhancing treatment efficacy.
By Therapeutic Area Analysis
The hematological disorders segment led the market in 2025, largely due to the high prevalence of leukemia, lymphoma, and myeloma, along with the established success of stem cell transplants and the increasing availability of off-the-shelf CAR-T therapies. Allogeneic stem cell transplantation is a well-established, effective, and occasionally curative treatment for hematological malignancies, bolstered by newer engineered allogeneic therapies. Innovations in cell engineering and the utilization of sources such as cord blood have improved the efficiency of therapy production, lowered costs and enhancing accessibility.
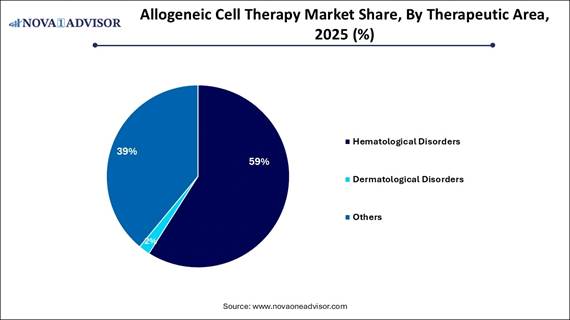
The dermatological disorders segment is forecasted to grow the fastest during the upcoming period. This growth is primarily driven by an increase in clinical trials focusing on skin diseases, wound healing, and anti-aging treatments. Advances in stem cell therapies, particularly those involving Mesenchymal Stem Cells, are vital for repairing or regenerating skin tissue. Furthermore, the demand for treatments for dermatological disorders is rapidly expanding, with projections indicating significant compound annual growth rates in the coming years, supported by a growing portfolio of products under development.
Allogeneic Cell Therapy Market Companies
· SSM Cardinal Glennon Children's Medical Center
· Duke University School of Medicine
· New York Blood Center
· Clinimmune Labs, University of Colorado Cord Blood Bank
· LifeSouth Community Blood Centers, Inc.
· Bloodworks Northwest
· JCR Pharmaceuticals Co., Ltd.
· Mallinckrodt Pharmaceuticals
· Takeda Pharmaceutical Company Limited
· STEMPEUTICS RESEARCH PVT LTD
· Biosolution Co., Ltd.
· MEDIPOST Co., Ltd.
Key Emerging Innovations in the Allogeneic Cell Therapy Market
|
Innovation |
Focus Area |
Example Product and Technology |
|
iPSC-Derived Cell Therapy |
Renewable, unlimited off-the-shelf cell sourcing (NK/T cells) |
FT596/FT819 (Fate Therapeutics) – iPSC-derived CAR-NK/CAR-T for B-cell malignancies |
|
Multiplex Genome Editing |
Simultaneous editing (CRISPR/TALEN) to knock out TCR/HLA to prevent GvHD and rejection |
UCART19 (Cellectis) – TCR/CD52 knocked out using TALEN, aiming at ALL |
|
Allogeneic CAR-NK Cells |
Utilizing Natural Killer cells for improved safety, lower GvHD risk, and solid tumor targeting |
AB-101 (Artiva Biotherapeutics) – NK cell therapy for non-Hodgkin's lymphoma |
|
Smart CARs and Logic-Gated Technology |
Enhancing precision by using dual receptors (Tmod) or cytokine engineering (IL-15) for better persistence |
SENTI-202 (Senti Bio) – Logic-gated CAR-NK cells for AML; Dual-targeted AlloCAR T (Allogene) |
|
In Vivo CAR-T Therapy |
Directing gene-editing tools via vectors to create CAR-T cells directly inside the patient's body |
Inceptor Bio's OUTLAST platform / Next-gen CAR-M or CAR-NK for solid tumors |
Major shifts in the Allogeneic Cell Therapy Market
· In March 2026, Lineage Cell Therapeutics, Inc. launched COR1, a new corneal endothelial cell (CEnC) therapy program in preclinical development for treating corneal endothelial disease, particularly Fuchs Endothelial Corneal Dystrophy and Bullous Keratopathy. Leveraging their AlloSCOPE™ platform, Lineage aims to produce consistent off-the-shelf CEnCs to address the global shortage of donor cells, as current cadaveric sources for developing a more effective product.
· In November 2025, Bharat Biotech International Ltd established Nucelion Therapeutics Pvt Ltd, a Contract Research, Development, and Manufacturing Organisation focused on cell and gene therapy. Located in Genome Valley, India, the facility is equipped for the comprehensive manufacturing of advanced therapies, aligning with US FDA and EMA regulations to serve global markets.
· In January 2026, Allogene Therapeutics, Inc. emphasized that the year would be pivotal for allogeneic CAR T therapies, with multiple clinical readouts planned to validate the scalability of off-the-shelf CAR T's in oncology and autoimmune disease. CEO David Chang highlighted their platform’s aim to ensure broad patient access and reliable treatment delivery, positioning it for expanded use across various cancers and conditions.
Related Report –
➡️ U.S. Mobile IV Hydration Therapy Market: Explore
the growing trend of on-demand hydration solutions
➡️ U.S. Aromatherapy Market: Understand the rising
demand for natural wellness therapies
➡️ Cell Therapy Human Raw Materials Market: Discover
the pivotal role of raw materials in advancing cell therapy
➡️ Advanced Therapy Medicinal Products Market: Unveil
the innovations in advanced therapy medicinal products
➡️ U.S. Cell Therapy Raw Materials Market: Analyze the
growth of raw materials essential for U.S. cell therapy
➡️ Stem Cell Therapy Market: Track the transformative
developments in stem cell therapy
➡️ U.S. Cell and Gene Therapy Market: Dive into the
expanding U.S. market for cell and gene therapies
➡️ U.S. Automated and Closed Cell Therapy Processing Systems
Market: Learn about automation's impact on cell therapy processing
systems
➡️ Hyperbaric Oxygen Therapy Devices Market: Explore
the growing use of hyperbaric oxygen therapy devices for wellness
➡️ RNA Therapy Clinical Trials Market: Discover the
potential of RNA therapy through clinical trials
➡️ Compression Therapy Market: Understand the
expanding market for compression therapy in healthcare
➡️ Cell and Gene Therapy Clinical Trials Market: Track
the clinical trials shaping the future of cell and gene therapies
➡️ CAR T-Cell Therapy Market: Investigate the breakthrough
CAR T-cell therapies in cancer treatment
➡️ Cancer Biological Therapy Market: Explore the
growing demand for cancer biological therapies
➡️ U.S. Cell and Gene Therapy Clinical Trials Market:
Gain insights into the clinical trials driving U.S. cell and gene therapies
➡️ Cell and Gene Therapy Contract Research Organizations Market:
Discover the role of CROs in advancing cell and gene therapy research
➡️ U.S. Physical Therapy Services Market: Explore the
increasing demand for physical therapy services in the U.S.
➡️ U.S. Compression Therapy Market: Uncover trends in
the U.S. compression therapy market for medical applications
➡️ U.S. Cell Therapy Market: Track the growth of the
U.S. cell therapy market and its key developments
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the allogeneic cell therapy market
By Therapy Type
- Stem Cell Therapies
- Hematopoietic Stem Cell Therapies
- Mesenchymal Stem Cell Therapies
- Non-stem Cell Therapies
- Keratinocytes & Fibroblast-based Therapies
- Others
By Therapeutic Area
- Hematological Disorders
- Dermatological Disorders
- Others
By Regional
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa (MEA)
Immediate Delivery Available | Buy This Premium Research https://www.novaoneadvisor.com/report/checkout/7933
About-Us
Nova One Advisor is a global leader in market intelligence and strategic consulting, committed to delivering deep, data-driven insights that power innovation and transformation across industries. With a sharp focus on the evolving landscape of life sciences, we specialize in navigating the complexities of cell and gene therapy, drug development, and the oncology market, enabling our clients to lead in some of the most revolutionary and high-impact areas of healthcare.
Our expertise spans the entire biotech and pharmaceutical value chain, empowering startups, global enterprises, investors, and research institutions that are pioneering the next generation of therapies in regenerative medicine, oncology, and precision medicine.
Web: https://www.novaoneadvisor.com/
Our Trusted Data Partners:
Nova One Advisor - Market Research Reports & Consulting Firm
Nova One Advisor offers comprehensive market research reports with in-depth industry analysis and market data.
Call us: +1 804 420 9370
Email: sales@novaoneadvisor.com
Web: https://www.novaoneadvisor.com/
You can place an order or ask any questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344


