United States
In a letter to FDA Commissioner Robert Califf, a House committee flagged several U.S. companies—including Eli Lilly and Pfizer—that have allegedly worked with the People’s Liberation Army on clinical trials in Xinjiang, raising ethical and intellectual property concerns.
As the pharma industry awaits congressional action on the bill, gaping holes in the domestic drug manufacturing ecosystem have never been clearer.
Experts say Novo Nordisk and Eli Lilly’s GLP-1 drugs are unlikely to reach more countries in the near term, but Sanofi’s diabetes treatment has gained ground globally.
Johnson & Johnson announced Tuesday that the FDA has approved Rybrevant with Lazcluze as a first-line chemotherapy-free treatment for patients with EGFR-mutated non-small cell lung cancer.
The retail pharmacy giant and the Biomedical Advanced Research and Development Authority hope to make decentralized clinical trials more accessible and representative of the U.S. population.
Two months after winning FDA approval in the same indication, AbbVie and Genmab on Monday secured the European Commission’s nod for its bispecific antibody Tepkinly for the most common type of low-grade non-Hodgkin lymphoma.
Eli Lilly released topline Phase III data on Tuesday showing that after more than three years of follow-up, pre-diabetic patients treated with tirzepatide were less likely to progress to type 2 diabetes.
Amgen’s Enbrel, Bristol Myers Squibb’s Eliquis and Johnson & Johnson’s Stelara will account for 51.4% of the Inflation Reduction Act’s drug price negotiation savings in 2026, according to the Brookings Institution.
The FDA on Monday only granted Liquidia’s Yutrepia tentative approval, keeping it off the U.S. market until after rival United Therapeutics’ exclusivity expires in May 2025.
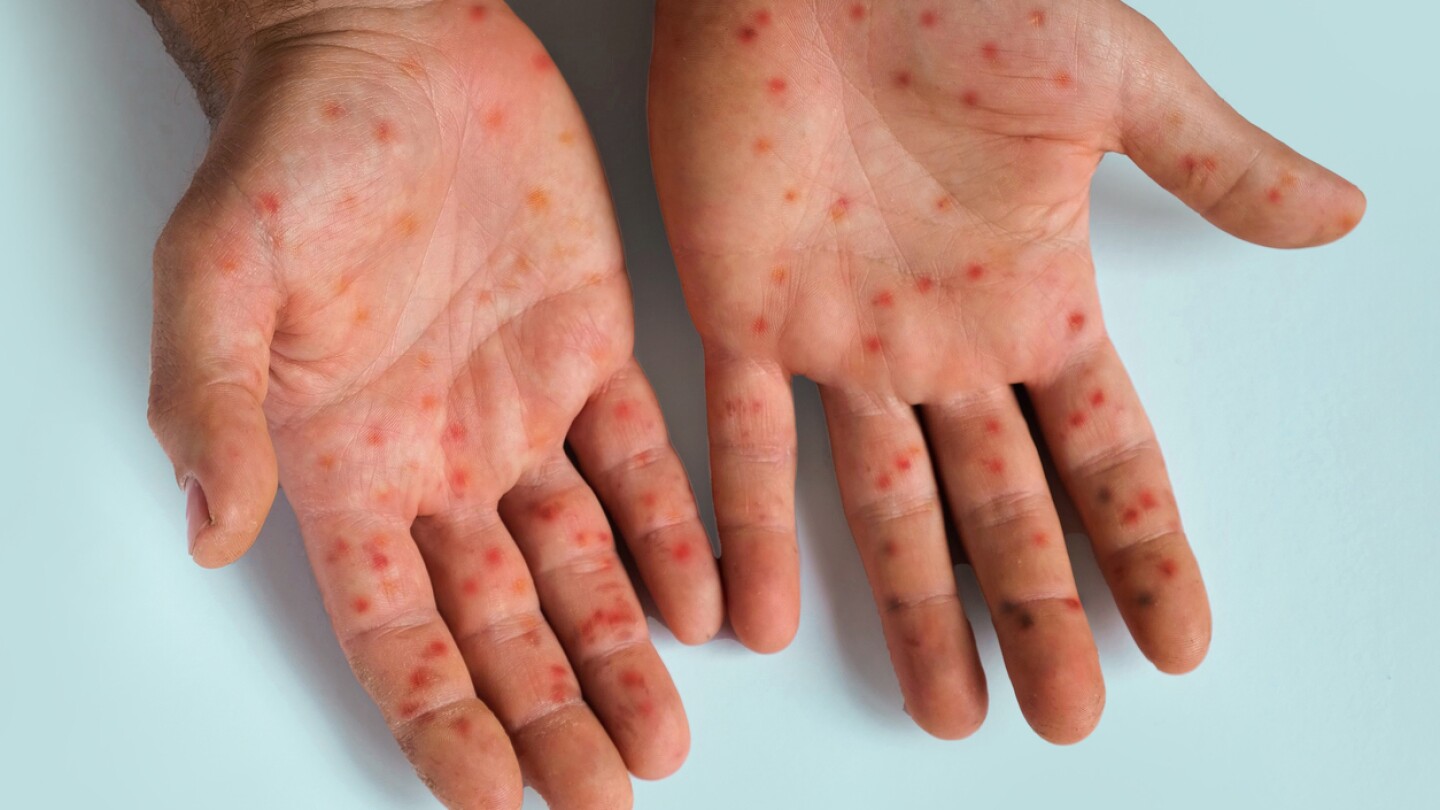
SIGA Technologies’ TPOXX did not outperform placebo at resolving lesions in patients with clade I mpox, the new strain that has spread through parts of Africa and is reaching beyond the continent.
PRESS RELEASES