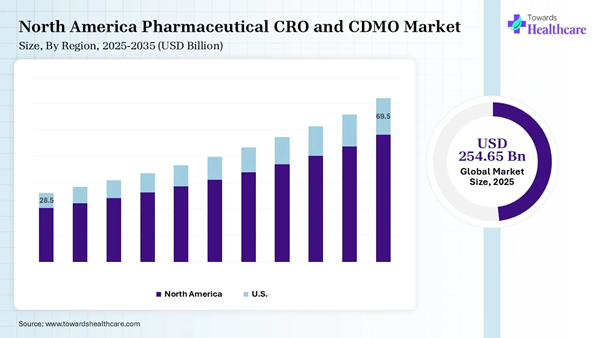
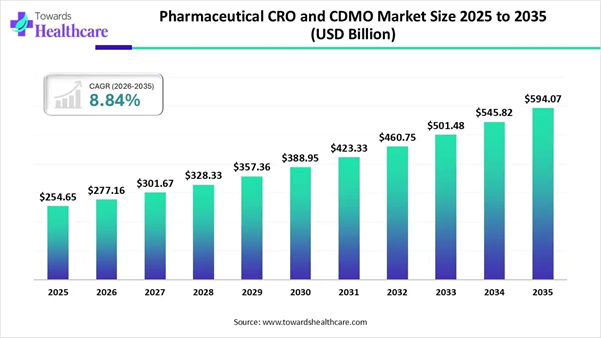
The global pharmaceutical CRO and CDMO market was valued at USD 254.65 billion in 2025 and is projected to grow from USD 277.16 billion in 2026 to approximately USD 594.07 billion by 2035, registering a CAGR of 8.84% during the forecast period (2026–2035).
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/6667
The contract research organization (CRO), contract development and manufacturing organization (CDMO), and contract manufacturing organization (CMO) are the cornerstones of biotechnology and pharmaceutical companies. The CDMO is pivotal in providing an integrated and end-to-end support across the lifecycle of a drug, and also offering clinical trial services. The CRO is essential to provide clinical research and clinical trial services to biopharmaceutical companies. The integration of CRO and CDMO for some programs helps to streamline operations.
The Pharmaceutical CRO and CDMO Market: Highlights
• Pharmaceutical CRO and CDMO market to crossed USD 277.16 billion by 2026.
• Market projected at USD 594.07 billion by 2035.
• CAGR of 8.84% expected in between 2026 to 2035.
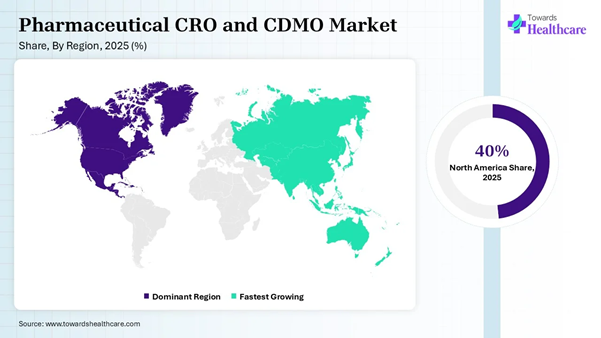
• North America dominated the global pharmaceutical CRO and CDMO market share by 40% in 2025.
• Asia Pacific is expected to be the fastest-growing region between 2026 and 2035.
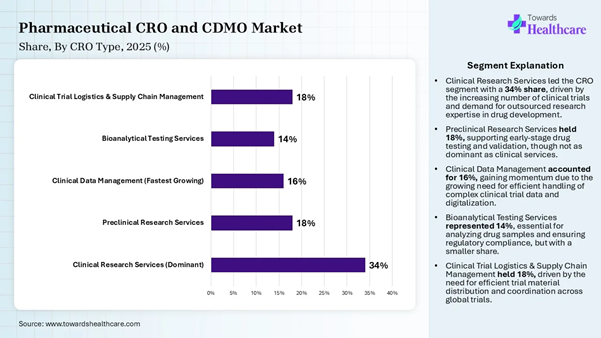
• By CRO type, the clinical research services (CRO) segment dominated the market share by 34% in 2025.
• By CRO type, the clinical data management (CRO) segment is expected to be the fastest growing during the forecast period.
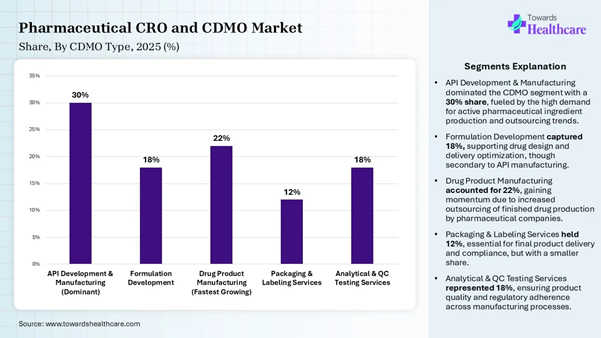
• By CDMO type, the API development and manufacturing (CDMO) segment dominated the market share by 30% in 2025.
• By CDMO type, the drug product manufacturing (CDMO) segment is expected to be the fastest-growing during the forecast period.
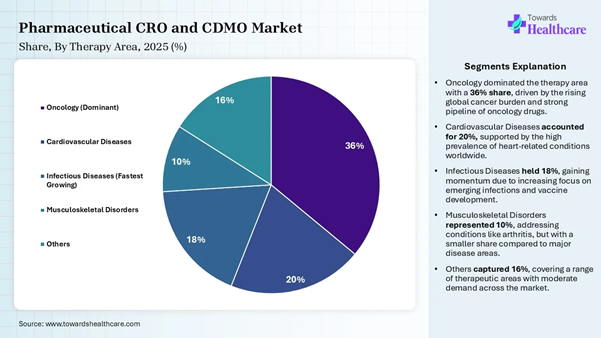
• By therapy area type, the oncology segment dominated the market share by 36% in 2025.
• By therapy area type, the infectious diseases segment is expected to be the fastest growing during the forecast period.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Market Overview
Clinical Research, Integrated Development, and Manufacturing Support
The pharmaceutical CRO and CDMO market is transitioning due to comprehensive support from CRO and CDMO in the drug development journey, from molecule to medicine. The CRO primarily focuses on clinical research and execution, while CDMO focuses on integrated development and manufacturing support. The CRO is the preferred choice of biotechnology and pharmaceutical companies when they need clinical trial services and coordination support.
On the other hand, CDMO is ideal when companies want integrated development and manufacturing under one partnership. The CRO supports trial planning, regulatory affairs, site selection, recruitment support, monitoring, data management, biostatistics, medical writing, and project management. Additionally, CDMO supports formulation development, regulatory support, clinical materials, packaging, supply chain, quality assurance, and tech transfer.
Integrated CRO and CDMO Services of Aurigene for Small and Large Molecules
Aurigene is one of the leading contract research, development, and manufacturing organizations, dedicated to supporting global pharmaceutical and biotechnology companies. It provides them with integrated services for small molecule discovery, small molecule CDMO, and biologics that further expand the pharmaceutical CRO and CDMO market. Accordingly, the featured offerings for small molecule discovery include integrated drug discovery, AI drug discovery, DNA encoded library screening services, CADD, and bioinformatics. Furthermore, small molecule CDMO services encompass offerings of peptides, high potency APIs, steroids, carbohydrates, mPEGs, formulations, and flow chemistry. Aurigene is leading in biologics offerings through antibody discovery, bispecifics, antibody drug conjugates, gene to protein, mAb, and protein therapeutics.
Importance of CRO and CDMO Model to Biopharma Companies
The biopharmaceutical companies across the pharmaceutical CRO and CDMO market are prioritizing a future-proofed supply chain management system, flexible manufacturing capacity, a wide range of capabilities, a robust governance framework, an integrated approach to drug discovery, and digitized or automated systems to drive more efficient and faster delivery. Syngene International Ltd. is one of the leading CRO/CDMO players with more than 420 active customers, 28 years of scientific experience, and more than 400 customer patents to its credit. It allows partnerships with biopharma companies to provide them with speed, cost efficiency, and reliability in their molecule discovery to commercialization.
Become a valued research partner with us - https://www.towardshealthcare.com/schedule-meeting
The Pharmaceutical CRO and CDMO Market: Regional Analysis
|
Region |
Share (%) |
|
North America (Dominant) |
40% |
|
Asia Pacific (Fastest Growing) |
27% |
|
Europe |
22% |
|
South America |
6% |
|
Middle East & Africa |
5% |
North America dominated the market share by
40% in 2025, owing to the supply chain resilience, complex biologics and cell
and gene therapies, decentralized clinical trials, and technological
integration. The CRO helps pharmaceutical and biotechnology players in this
region to coordinate and manage a clinical trial and its progress, which
further decides the development and manufacturing of a drug. The
pharmaceutical, biotechnology, and medical
device companies prefer outsourcing a wide range of clinical trial
services. These services enable them to utilize capabilities, knowledge, resources,
and infrastructure while working on important tasks. U.S. Market Analysis The pharmaceutical CRO and CDMO market in
the U.S. is transformed due to a surging demand for obesity and diabetes
medications, which has created a massive need for peptide synthesis services
and sterile fill-finish. The National Priority Voucher program introduced
incentives for domestic manufacturing and accelerated regulatory reviews. Asia Pacific is expected to grow at the
fastest rate in the market during the forecast period due to access to large
and diverse patient pools, significant investments in state-of-the-art good
manufacturing practice (GMP) facilities, and specialized manufacturing for
biologics and sterile injectables. The government initiatives aim to position
the Asia Pacific region as a global biopharma hub by offering streamlined regulations,
financial incentives, and infrastructure development. India Market Trends The pharmaceutical CRO and CDMO market in
India is witnessing a large number of U.S. FDA-compliant facilities, and
incentives for high-value R&D and manufacturing of complex biosimilars and
APIs. The Production Linked Incentive (PLI) scheme directly impacts CDMO
capacities, while the BioE3 policy, approved by the Union Cabinet, boosts
high-performance biomanufacturing. Get the latest insights on life science
industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership The Pharmaceutical CRO and CDMO Market:
Segmentation Analysis CRO Type Insights Segment Share (%) Clinical Research Services
(Dominant) 34% Preclinical Research Services 18% Clinical Data Management
(Fastest Growing) 16% Bioanalytical Testing
Services 14% Clinical Trial Logistics
& Supply Chain Management 18%
The clinical research services segment
dominated the market share by 34% in 2025, owing to the increased focus on
antibody drug conjugates, biologics, cell and gene therapies, specialized
injectables, and radiopharmaceuticals.
Large-scale investments are seen in new and expanded manufacturing sites across
regions like Europe, Asia Pacific, and North America. The companies are moving
towards leveraging talent, expertise, technologies, and facilities to expand
their R&D footprint. The clinical data management segment is
expected to be the fastest-growing in the pharmaceutical CRO and CDMO market
during the forecast period due to the wide use of clinical data management
systems to enable the source collection and management of patient data. The
clinical data management organization plays a pivotal role in designing case
report forms for the electronic data capture system. They also support medical
safety reviews and central monitoring as part of a risk-based quality
management system. CDMO Type Insights Segment Share (%) API Development &
Manufacturing (Dominant) 30% Formulation Development 18% Drug Product Manufacturing
(Fastest Growing) 22% Packaging & Labeling
Services 12% Analytical & QC Testing
Services 18%
The API development and manufacturing
segment dominated the market share by 30% in 2025, owing to the importance of
API in the pharma industry, which impacts the efficacy of the drug and
maintains the quality and safety of the drug. These industrial stages ensure
adherence of each drug to the regulations of the authority. API development
empowers innovation in the pharma industry by creating new-generation molecules
and targeted
therapy drugs for specialized treatments. The drug product manufacturing segment is
expected to be the fastest-growing in the pharmaceutical CRO and CDMO market
during the forecast period due to the leading role of CROs in early research
and clinical management. The integration of CDMOs is increasing as long-term
partners for the entire commercial lifecycle of the drug. There is a rising
demand for manufacturing biologics, highly potent APIs, and cell and gene
therapies. Therapy Area Type Insights Segment Share (%) Oncology (Dominant) 36% Cardiovascular Diseases 20% Infectious Diseases (Fastest
Growing) 18% Musculoskeletal Disorders 10% Others 16%
The oncology segment dominated the market
share by 36% in 2025, owing to the major assistance of CDMO for the oncology
sector in providing pharma brands and companies with compliant manufacturing of
cancer drugs. Modern CDMOs are flexible and adapt to the latest trends
in oncology CDMO. They adapt to managing high potency APIs, outsourcing
complex drug production, and supporting innovative drug formats. The infectious diseases segment is expected
to be the fastest-growing in the pharmaceutical CRO and CDMO market during the
forecast period due to the expanding role of CROs and CDMOs in addressing the
complexities of antimicrobial resistance, rapidly evolving pathogens, and the
need for global pandemic preparedness. CROs contribute to managing the research
and clinical trial lifecycle to ensure the safety and efficacy of a drug. The
CROs and CDMOs provide advanced biosafety and microbiology expertise needed for
handling infectious agents. Access our comprehensive healthcare
dashboard for detailed market insights, segment breakdowns, regional
performance, and company profiles: https://www.towardshealthcare.com/access-dashboard The Pharmaceutical CRO and CDMO Market
Companies • IQVIA • ICON Plc • Parexel • Charles River Laboratories • Medpace • Lonza Group • Samsung Biologics • Catalent • Boehringer Ingelheim • Thermo Fisher Scientific • Fujifilm Diosynth Biotechnologies • Siegfried Holding AG • Fortrea Segments Covered in the Report By CRO • Clinical Research Services (CRO) • Preclinical Research Services (CRO) • Clinical Data Management (CRO) • Bioanalytical Testing Services (CRO) • Clinical Trial Logistics and Supply Chain
Management (CRO) By CDMO • Formulation Development (CDMO) • API Development and Manufacturing (CDMO) • Drug Product Manufacturing (CDMO) • Packaging and Labeling Services (CDMO) • Analytical and QC Testing Services (CDMO) By Therapy Area • Oncology • Cardiovascular Diseases • Infectious Diseases • Musculoskeletal Disorders • Others By Region • North America • South America • Europe • Asia Pacific • MEA The Complete Study is Now Available for
Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/checkout/6667 About Us Towards Healthcare is a leading global provider of technological solutions, clinical
research services, and advanced analytics, with a strong
emphasis on life science research. Dedicated to
advancing innovation in the life sciences sector, we build strategic
partnerships that generate actionable insights and transformative
breakthroughs. As a global strategy consulting firm, we empower life science
leaders to gain a competitive edge, drive research excellence, and accelerate
sustainable growth. You can place an order or ask any
questions, please feel free to contact us at sales@towardshealthcare.com Europe Region: +44 778 256 0738 North America Region: +1 8044 4193 44 APAC Region:
+91 9356 9282 04 Web: https://www.towardshealthcare.com Our Trusted Data Partners Precedence
Research | Towards Packaging | Towards
Food and Beverages | Towards
Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest Browse More Insights of Towards
Healthcare: ➡️ The oncolytic
virus CDMO services market size reached US$ 419.51 million in 2025 and
is anticipate to increase to US$ 483.49 million in 2026. By 2035, the
market is forecasted to achieve a value of around US$ 1734.41 million, growing
at a CAGR of 15.25%. ➡️ The global biologics
CDMO market size is calculated at USD 22 billion in 2024, grew to USD
25.41 billion in 2025, and is projected to reach around USD 92.79 billion by
2034. The market is expanding at a CAGR of 15.48% between 2025 and 2034. ➡️ The U.S.
pharmaceutical CDMO market size is calculated at USD 39.13 billion in
2025, grew to USD 41.65 billion in 2026, and is projected to reach around USD
72.98 billion by 2035. The market is expanding at a CAGR of 6.43% between 2026
and 2035. ➡️ The active
pharmaceutical ingredients CDMO market size is calculated at USD 127.45
billion in 2024, grew to USD 136.92 billion in 2025, and is projected to reach
around USD 260.98 billion by 2034. The market is expanding at a CAGR of 7.43%
between 2025 and 2034. ➡️ The global healthcare
CDMO market size was estimated at USD 306.3 billion in 2025 and is
predicted to increase from USD 337.3 billion in 2026 to approximately USD
803.17 billion by 2035, expanding at a CAGR of 10.12% from 2026 to 2035. ➡️ The global mRNA
CDMO service market size was estimated at USD 5.53 billion in 2025 and
is predicted to increase from USD 5.94 billion in 2026 to approximately USD
11.25 billion by 2035, expanding at a CAGR of 7.36% from 2026 to 2035. ➡️ The global AAV
vector CDMO services market size is calculated at USD 1.24 billion in
2024, grew to USD 1.43 billion in 2025, and is projected to reach around USD
5.14 billion by 2034. The market is expanding at a CAGR of 15.24% between 2025
and 2034. ➡️ The global small
molecule CDMO market size is calculated at US$ 72.81 in 2024, grew to
US$ 78.01 billion in 2025, and is projected to reach around US$ 145.12 billion
by 2034. The market is expanding at a CAGR of 7.14% between 2025 and 2034. ➡️ The global pharmaceutical
CDMO market size was estimated at US$ 167.96 billion in 2025 and is
projected to grow to US$ 337.89 billion by 2035, rising at a compound annual
growth rate (CAGR) of 7.24% from 2026 to 2035. ➡️ The global biotechnology
CMO and CDMO market size is estimated at US$ 67.25 billion in 2024, is
projected to grow to US$ 74.01 billion in 2025, and is expected to reach around
US$ 199.67 billion by 2034. The market is projected to expand at a CAGR of
11.54% between 2025 and 2034.