According to Nova One Advisor, the global mRNA therapeutics market size was valued at USD 17.25 Billion in 2025 and is poised to grow from USD 20.20 Billion in 2026 to USD 83.49 Billion by 2035, at a CAGR of 17.08% during the forecast period (2026-2035).
Across the globe, the shift from pandemic-focused vaccines to personalized medicines in oncology, infectious diseases, and rare disorders is supported by significant investment in next-generation delivery systems like self-amplifying RNA (saRNA) and advanced lipid nanoparticles (LNPs). To tackle high costs and regulatory barriers, the industry is moving toward automated, continuous-flow manufacturing in modular facilities. Additionally, the integration of AI-driven digital twins and predictive modeling is optimizing mRNA design and validation, leading to increased efficiency and faster time-to-market for personalized therapeutic proteins and cancer vaccines.
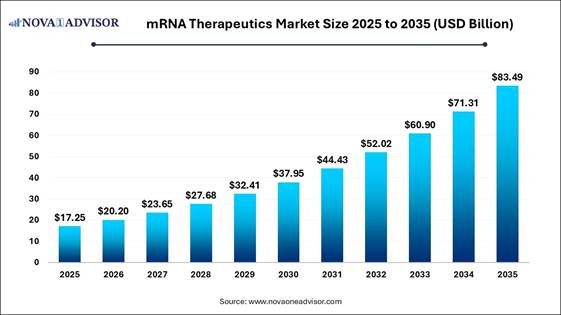
Key Takeaways
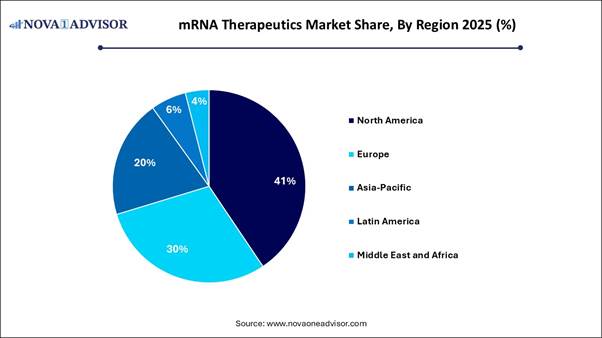
· North America dominated with the largest market share in 2025.
· Asia Pacific is anticipated to have the fastest growth with a notable CAGR during the forecast period.
· By application, the oncology segment contributed the highest market share in 2025.
· By application, the rare genetic diseases segment is growing at a strong CAGR between 2026 and 2035.
· By type, the prophylactic segment held a major market share in 2025.
· By type, the therapeutic segment is expected to expand at a notable CAGR from 2026 to 2035.
· By end-use, the hospitals and clinics segment captured the highest market share in 2025.
· By end-use, the research organizations segment is poised to grow at a healthy CAGR between 2026 and 2035.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/6847
What are mRNA Therapeutics?
mRNA therapeutics are medicines that make use of ribonucleic acid molecules to treat or prevent disease. They do that by influencing the flow of genetic information. Unlike traditional drugs that target protein downstream, RNA therapies work upstream, which is at the level of genetic messaging. It mimics the structure of naturally occurring transcripts, complete with a cap, untranslated regions, and a poly(A) tail. Once encapsulated in LNPs and delivered into the cytoplasm, the mRNA is read by ribosomes to produce the encoded protein. Because the RNA never enters the nucleus, there is no risk of altering the genome.
The mRNA market has experienced significant growth and transformation over the recent years, driven by advancements in biotechnology and the successful application of mRNA technology in vaccine development, especially during the Covid-19 pandemic.
Market Overview: The New Frontier in Personalized Cancer Care
The mRNA therapeutics market involves designing synthetic mRNA to instruct cells to produce therapeutic proteins, targeting diseases such as cancer and infectious diseases. Rapid growth is driven by advances in lipid nanoparticle delivery and a strategic focus on oncology and rare genetic disorders. While prophylactic vaccines still hold a significant share, the market is diversifying into personalized cancer immunotherapies and protein replacement therapies, supported by a robust biotech infrastructure. Key growth drivers include increased investments, strategic collaborations, and development of next-generation, self-amplifying platforms.
Personalized Neoantigen Vaccines: Major Potential
The mRNA technology landscape is shifting toward a versatile, plug-and-play platform enabling quick, personalized, and highly stable therapies for cancer, especially via patient-specific neoantigen vaccines, and rare genetic disorders. With advancements in LNPs and personalized oncology, therapeutic mRNA applications, such as in influenza and respiratory syncytial virus, are expected to surpass the growth of prophylactic COVID-19 vaccines, expanding the overall personalized mRNA therapeutics market.
High Costs and Cold Chain Constraints: Major Limitations
Major barriers include complex manufacturing processes requiring multi-step purification and costly cGMP-grade reagents, along with severe stability issues that demand expensive ultra-cold chain logistics, limiting accessibility. Beyond production and storage challenges, regulatory uncertainties and patent disputes over LNPs delay market entry. Technical hurdles related to safety, such as off-target effects and managing adverse immune responses from long-term, repeated dosing, also pose significant challenges.
Immediate Delivery is Available | Get Full Report Access@ https://www.novaoneadvisor.com/report/checkout/6847
mRNA Therapeutics Market Report Scope
|
Report Attribute |
Details |
|
Market Size in 2026 |
USD 20.2 Billion |
|
Market Size by 2035 |
USD 83.49 Billion |
|
Growth Rate From 2026 to 2035 |
CAGR of 17.06% |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2035 |
|
Segments Covered |
Application, Type, End-use, Region |
|
Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
|
Report Coverage |
Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
|
Key Companies Profiled |
Moderna Inc.; BioNTech SE; CureVac N.V.; Arcturus Therapeutics; Sanofi; GSK Plc.; Argos Therapeutics Inc.; Ethris; Pfizer Inc.; AstraZeneca |
You can place an order or ask any questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344
mRNA Therapeutics Market: Segmentation Analysis
By Application Analysis
The oncology segment dominated the market in 2025, primarily due to its transformation from a high-potential research area into the largest revenue-generating application. mRNA technology facilitates the rapid, patient-specific design of vaccines that prompt the immune system to recognize tumor-specific neoantigens, resulting in targeted attacks on cancer cells. The oncology pipeline for mRNA therapeutics is rapidly expanding, with numerous ongoing clinical trials in Phase II and III targeting melanoma, lung, and pancreatic cancers.
The segment for rare genetic diseases is expected to experience the fastest growth during the forecast period. This growth is driven by the technology's ability to deliver instructions for producing functional proteins, addressing the root causes of enzyme deficiencies. Advances in mRNA modification and delivery systems have improved the stability and efficiency of these therapies, enabling the production of specific functional proteins in the body to treat diseases caused by absent or faulty enzymes.
By Type Analysis
The prophylactic segment led the market in 2025, primarily due to the extensive deployment of COVID-19 vaccines and the expansion of mRNA technology to other infectious diseases. Products like Comirnaty and Spikevax continue to generate substantial revenue, maintaining market dominance through regular booster doses and adapted formulations for new variants. The technology's ability to quickly design, modify, and produce vaccines provides a competitive advantage over traditional methods, making it ideal for pandemic preparedness and seasonal virus management.
The therapeutic segment is projected to grow the fastest during the forecast period. This growth is mainly fueled by the expanding pipeline of personalized mRNA cancer vaccines targeting diseases such as melanoma, lung, and pancreatic cancers. mRNA technology is increasingly utilized for protein replacement therapies to treat rare diseases, attracting significant investment in research and development, and obtaining valuable Orphan Drug Designations. These advancements enhance the stability and delivery efficiency of mRNA therapeutics while reducing toxicity and increasing efficacy.
By End-use Analysis
The hospitals and clinics segment dominated the market in 2025, mainly due to their role as the primary distribution hub for COVID-19 vaccines and emerging cancer immunotherapies. Hospitals have the necessary advanced infrastructure and trained professionals to manage mRNA therapies and monitor patients effectively. The expansion of mRNA applications into oncology and chronic disease treatments drives significant patient traffic to hospitals. The rising incidence of infectious diseases and cancer guarantees a steady demand for new mRNA-based therapeutics.
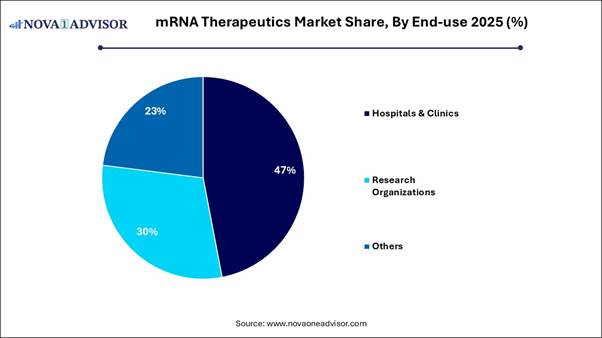
The research organizations segment is projected to experience the fastest growth during the forecast period. This growth is primarily driven by increasing public and private funding for research and development, heightened preclinical studies, and partnerships with CDMOs. Growing funding from government agencies and private investors targeting early-stage mRNA research, particularly in oncology and rare genetic disorders, is accelerating this segment's growth. Research centers are increasingly focusing on non-pandemic applications, such as personalized cancer vaccines and immunotherapies, which require extensive early-stage investigation.
Buy Now Full Report: https://www.novaoneadvisor.com/report/checkout/6847
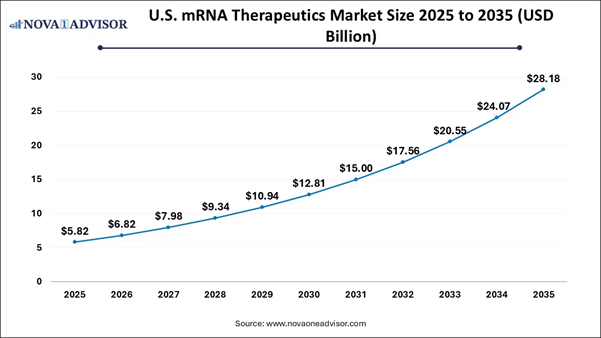
What is the Size of the U.S. mRNA Therapeutics Market?
The U.S. mRNA therapeutics market size is calculated at USD 5.82 billion in 2025 and is expected to reach nearly USD 28.18 billion in 2035, accelerating at a strong CAGR of 15.42% between 2026 and 2035.
mRNA Therapeutics Market: Regional Analysis
North America led the market in 2025, mainly driven by substantial research investments, a strong presence of industry leaders, and favorable government policies. The region hosts pioneering companies like Moderna, Inc., Pfizer Inc., and BioNTech, which dominate the commercial landscape. The FDA’s Emergency Use Authorization process accelerated approvals, setting a record for rapid deployment of new mRNA therapies. The U.S. government continues to support this field through initiatives like the NIH and Operation Warp Speed, emphasizing mRNA research and manufacturing infrastructure.
As the global leader, the U.S. mRNA therapeutics market is poised for explosive growth, fueled by large investments, top-tier infrastructure, and major players like Moderna and Pfizer. Fast-tracked FDA approvals, along with a focus on personalized oncology, vaccines, and rare diseases reinforce America’s dominance in pioneering the future of rapid and transformative medicine.
Canada is swiftly emerging as a global mRNA hub, leveraging strategic government backing and partnerships, such as Moderna’s new Laval facility, to strengthen its domestic fill-and-finish capacity and enhance supply security. The country is shifting from an importer to an innovator, concentrating on infectious diseases and pioneering cancer therapies to secure a central role in mRNA medicine’s future.
· In September 2025, Moderna officially launched its state-of-the-art Laval, Quebec facility, delivering the first domestically produced mRNA Spikevax vaccines. This milestone establishes a fully Canadian end-to-end supply chain, boosting national health security with locally manufactured medicines.
Asia Pacific is expected to experience the fastest growth during the forecast period. This expansion is largely fueled by significant government investments in biotechnology, a large patient population, rapid scaling of manufacturing capabilities, and increased clinical research activities in countries like China, Japan, and India. Governments in China and Japan are investing heavily in mRNA research, development, and infrastructure to achieve supply chain independence. The rising incidence of chronic and infectious diseases is driving demand for advanced, personalized therapies solutions.
India is rapidly emerging as a global powerhouse in the mRNA therapeutics market, driven by its success in developing indigenous, cost-effective mRNA platforms and a rising demand for personalized medicine. The country has achieved comprehensive capabilities, spanning research and development to manufacturing, which reduces reliance on imports and establishes it as a crucial hub for next-generation vaccines and therapies targeting cancers and infectious diseases.
· In March 2026, the Biopharma SHAKTI initiative is a landmark ₹10,000-crore, five-year national program designed to propel India from being a generics-only manufacturer to a global leader in biologics and biosimilars.
Meanwhile, China is rapidly asserting dominance over the Asian mRNA landscape by transitioning from a manufacturing base to a leader in research and development. This shift has led to a surge in clinical trials focused on oncology and infectious diseases. With significant investment in proprietary LNP technology, China is moving beyond COVID-19 to secure a leading position in specialized markets.
mRNA Therapeutics Market Companies
· CureVac N.V.
· Arcturus Therapeutics
· Sanofi
· GSK Plc.
· Ethris
Emerging Innovations in the mRNA Therapeutics
|
Innovation |
Description |
Primary Focus Area |
|
Self-Amplifying mRNA (saRNA) |
mRNA that replicates inside the cell, allowing lower, more durable dosing. Example: Arcturus Therapeutics’ ARCT-154 (COVID-19/Influenza). |
Infectious Disease and Long-term Prophylaxis |
|
Circular RNA (circRNA) |
A covalently closed loop structure that offers greater stability and longer protein expression than linear mRNA. Example: Orna Therapeutics’ oRNA platforms. |
Rare Genetic Diseases and Oncology |
|
Next-Gen LNP Delivery |
Advanced Lipid Nanoparticles (LNPs) designed for specific tissue targeting and non-injectable delivery. Example: Ethris’ inhalable LNPs (SNIM®RNA). |
Personalized Immuno-Oncology |
|
Personalized Neoantigen Vaccines |
Tailored mRNA vaccines using AI/ML to target unique mutations in a patient's tumor. Example: BioNTech's BNT122 for cancer. |
Personalized Immuno-Oncology |
|
mRNA-Encoded Antibodies |
Delivering mRNA to instruct cells to produce therapeutic antibodies in vivo. Example: BioNTech’s BNT142 (anti-CLDN6 bispecific). |
Oncology and Protein Replacement |
Major Shifts in the mRNA Therapeutics Market
· In August 2025, Pfizer Inc. and BioNTech SE announced that the FDA approved their LP.8.1-adapted monovalent COVID-19 vaccine (COMIRNATY® LP.8.1) for adults 65 and older, and individuals 5 to 64 with high-risk underlying conditions. This approval is supported by clinical trial data, including data for children aged 5 to 11. The new vaccine generates improved immune responses against various SARS-CoV-2 sublineages and is set to be shipped to pharmacies and clinics.
· In October 2025, Novartis revealed its plans to acquire Avidity Biosciences, following the separation of Avidity’s early-stage cardiology programs. Avidity focuses on RNA therapeutics for genetic neuromuscular diseases and brings a differentiated RNA-targeting delivery platform to Novartis, enhancing its neuroscience strategy.
· In August 2025, Raina Biosciences published a study highlighting its generative AI platform, GEMORNA, which designs mRNA sequences with enhanced properties. CEO Jicong Cao reported significant improvements in drug expression and efficacy, emphasizing the platform's potential to transform mRNA therapeutics and its versatility across various applications.
Related Report –
➡️ U.S. mRNA Synthesis Raw Materials Market: Examine how increasing mRNA-based research and vaccine production are driving demand for high-quality raw materials
➡️ U.S. mRNA Therapeutics Market: Explore rapid advancements in mRNA-based treatments transforming the landscape of precision medicine
➡️ mRNA Synthesis & Manufacturing Market: Analyze the expansion of scalable manufacturing technologies supporting global mRNA pipeline growth
➡️ mRNA Therapeutics Contract Development & Manufacturing Organization Market: Discover how outsourcing trends are accelerating innovation and production efficiency in mRNA therapeutics
➡️ mRNA Synthesis Raw Materials Market: Gain insights into supply chain dynamics and critical inputs powering mRNA production worldwide
➡️ Non-small Cell Lung Cancer Therapeutics Market: Understand how targeted therapies and immunotherapies reshaping treatment outcomes in lung cancer are.
➡️ Geriatric Medicine Market: Explore rising healthcare demand driven by aging populations and chronic disease management needs
➡️ Monoclonal Antibody Therapeutics Market: Track the growing adoption of biologics in treating cancer, autoimmune disorders, and infectious diseases
➡️ U.S. Digital Therapeutics Market: Analyze how software-based therapies are revolutionizing disease management and patient engagement
➡️ Asthma Therapeutics Market: Discover innovations in biologics and inhalation therapies improving respiratory care outcomes
➡️ RNA Therapeutics Market: Examine the expanding role of RNA-based drugs in treating rare and chronic diseases
➡️ Leukemia Therapeutics Market: Gain insights into evolving treatment approaches including targeted therapies and cell-based treatments
➡️ U.S. Antisense And RNAi Therapeutics Market: Explore gene-silencing technologies driving breakthroughs in genetic and rare disease therapies
➡️ Lymphoma Therapeutics Market: Understand how immunotherapy and precision medicine are transforming lymphoma treatment strategies
➡️ Natural Killer Cells Therapeutics Market: Analyze the rise of cell-based immunotherapies leveraging NK cells for cancer treatment
➡️ Cystic Fibrosis Therapeutics Market: Discover advancements in CFTR modulators and personalized therapies improving patient outcomes
➡️ Anti-Inflammatory Therapeutics Market: Examine growing demand for treatments targeting chronic inflammation across multiple disease areas
mRNA Therapeutics Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the mRNA Therapeutics market.
By Application
· Infectious Diseases
· Oncology
· Rare Genetic Diseases
· Respiratory Diseases
· Others
By Type
· Prophylactic
· Therapeutic
By End-use
· Hospitals & Clinics
· Research Organizations
· Others
By Region
· North America
· Europe
· Asia-Pacific
· Latin America
· Middle East & Africa (MEA)
Immediate Delivery Available | Buy This Premium Research https://www.novaoneadvisor.com/report/checkout/6847
About-Us
Nova One Advisor is a global leader in market intelligence and strategic consulting, committed to delivering deep, data-driven insights that power innovation and transformation across industries. With a sharp focus on the evolving landscape of life sciences, we specialize in navigating the complexities of cell and gene therapy, drug development, and the oncology market, enabling our clients to lead in some of the most revolutionary and high-impact areas of healthcare.
Our expertise spans the entire biotech and pharmaceutical value chain, empowering startups, global enterprises, investors, and research institutions that are pioneering the next generation of therapies in regenerative medicine, oncology, and precision medicine.
Web: https://www.novaoneadvisor.com/
Our Trusted Data Partners:
Nova One Advisor - Market Research Reports & Consulting Firm
Nova One Advisor offers comprehensive market research reports with in-depth industry analysis and market data.
Call us: +1 804 420 9370
Email: sales@novaoneadvisor.com
Web: https://www.novaoneadvisor.com/
You can place an order or ask any questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344


