Phase 2
The Swiss pharma has canceled a second mid-stage trial for its investigational schizophrenia drug ralmitaront, as the competition shows promise in the space.
Data from a Phase III trial of apraglutide for a type of short bowel syndrome with intestinal failure is expected by the end of the year. It is also in a Phase II study for acute graft-versus-host disease.
The 61-patient study met the primary endpoint of improved physiologic liver function and secondary endpoints, which included established non-alcoholic steatohepatitis biomarkers, Hepion reported Monday.
Fenebrutinib in a mid-phase trial reduced brain legions in relapsing forms of multiple sclerosis, the company reported. It is the only reversible BTK inhibitor currently in Phase III MS trials.
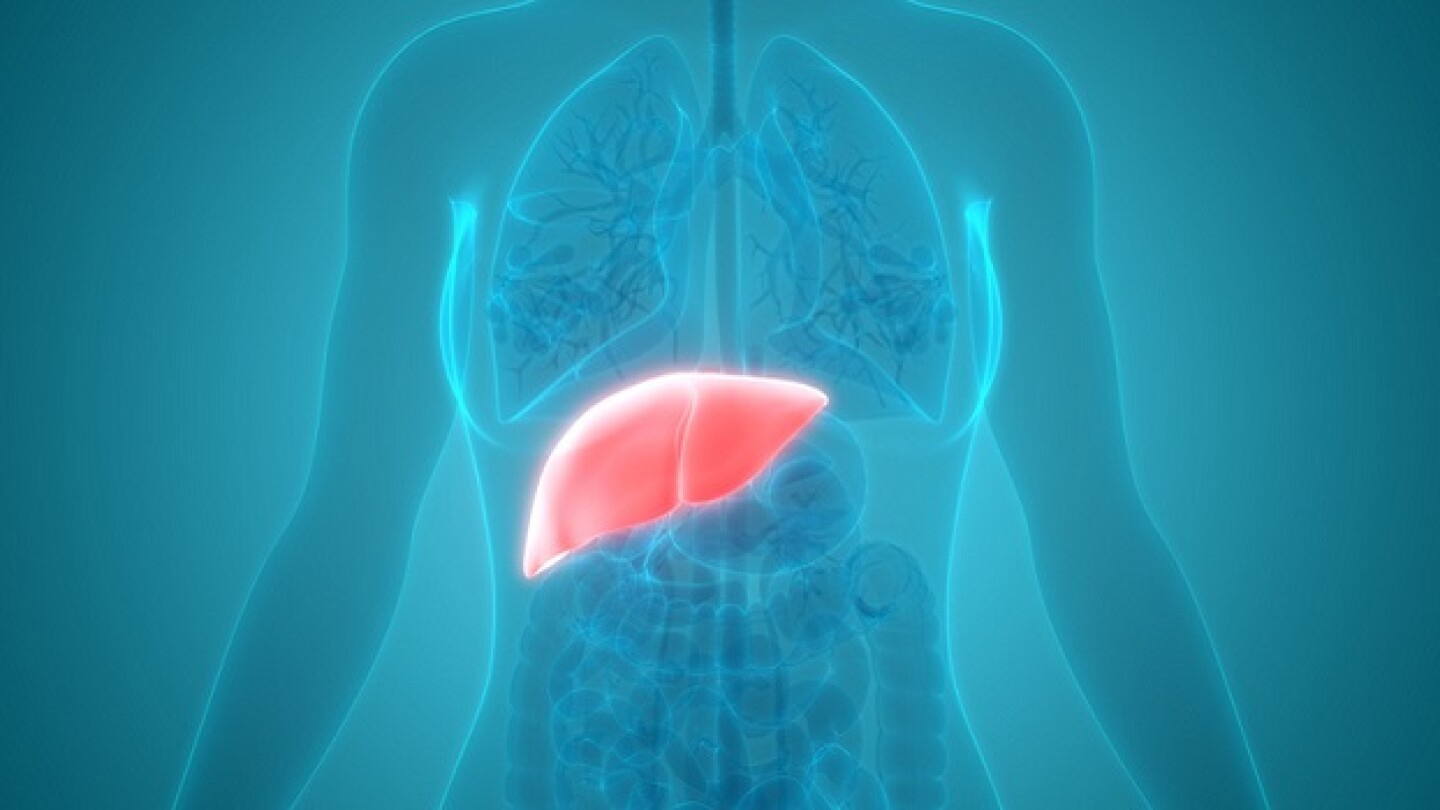
Results from Phase IIb VOYAGE study showed Viking’s oral thyroid beta receptor agonist cut liver fat content by up to 51% in confirmed non-alcoholic steatohepatitis patients.
The approval of Reata Pharmaceuticals’ Skyclarys for Friedreich’s ataxia highlights progress being made in the treatment of rare mitochondrial diseases.
Developers of psychedelics-based therapies say the industry is poised to explode, with several reporting strong clinical trial results and the FDA granting breakthrough status for two hallucinogenic drugs.
The layoffs come as the company posts nearly $300 million in net losses and just over $80 million in revenue during the first quarter of 2023.
The deal with BlissBio is the latest in a cascade of multi-million- and billion-dollar acquisitions and collaborations centered on antibody-drug conjugates.
The FDA’s new guidance on decentralized clinical trials clarifies how investigators and trial sponsors can use digital health technologies and involve primary healthcare practitioners.
PRESS RELEASES