Infectious disease
President Biden is expected to be briefed on the lab leak theory conducted over the last 90 days. Very few expect solid results, although this might provide direction for more lines of inquiry.
The new authorization is expected to be the final reassurance for many of those who had hesitated when the vaccine was approved under emergency use only.
An oral version of remdesivir is being developed by scientists at the University of California San Diego to combat the SARS-CoV-2 virus early in its infection cycle.
While last flu season saw fears of a “twindemic” go largely unfounded, there are fears that recent lockdowns could also lead to a lack of natural immunity against the flu.
Research presented earlier this month discussed a possible link between COVID-19 and long-term cognitive deficits and how it may accelerate the pathology and symptoms of Alzheimer’s disease.
Many expect the full approval will lead to vaccine mandates, as that was one of the obstacles to it for many organizations.
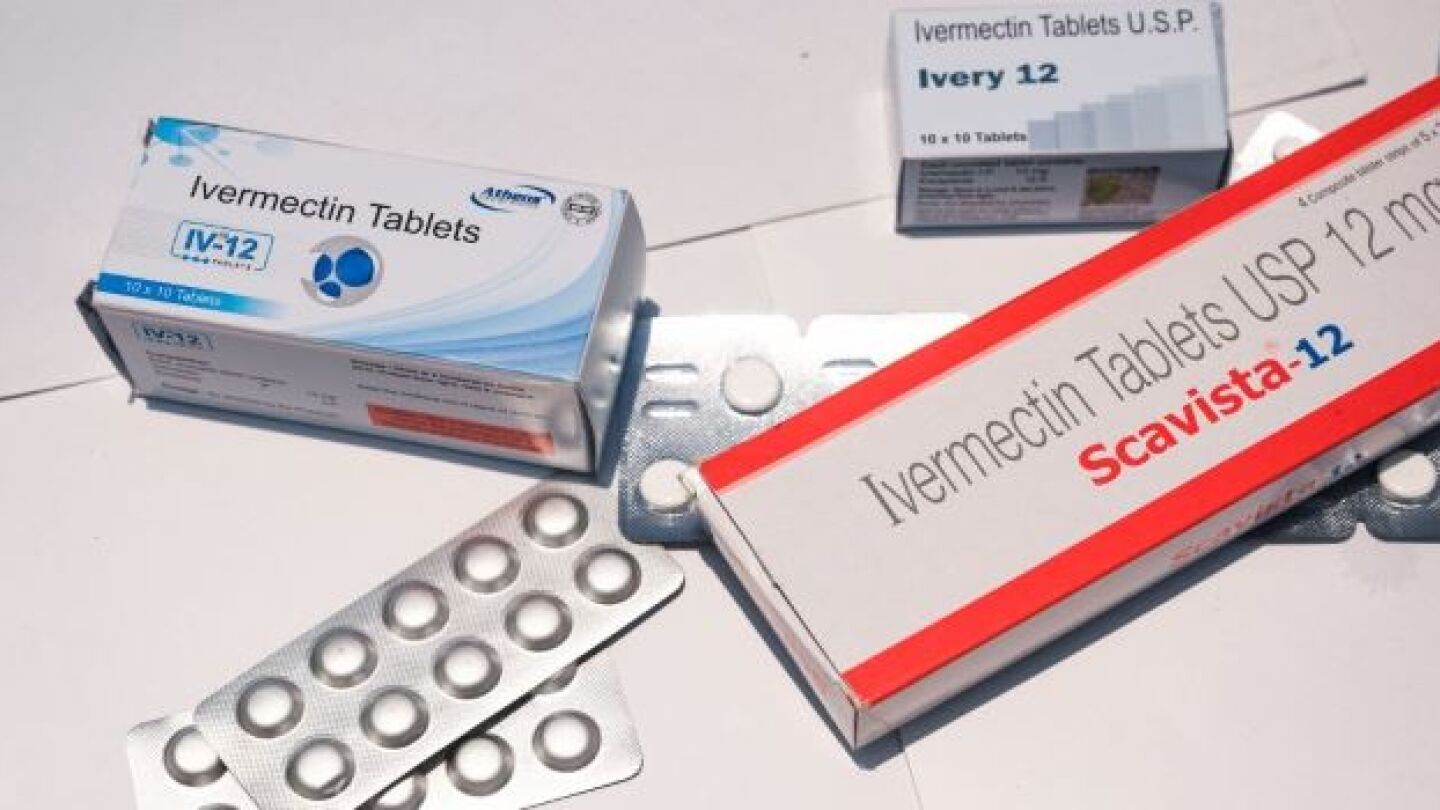
“You are not a horse. You are not a cow. Seriously, y’all. Stop it.” This is the FDA’s recently tweeted advice to Americans seeking out alternative, unapproved treatments for COVID-19.
Starpharma’s Viraleze has reduced the viral load by over 99.9% when it was administered nasally into the trachea and lungs of animals who were positive with the virus, compared to saline control.
Research emerging from Israel identified eight already-approved compounds that inhibit the activity of the E-protein channels in the virus’s membrane.
As the delta variant continues to contribute to the majority of COVID-19 cases worldwide, many patients are reporting delta variant symptoms which is a great concern.
PRESS RELEASES