Phase 2
Full data for Eli Lilly’s Phase III TRAILBLAZER-ALZ 2 study, presented Monday at the 2023 Alzheimer’s Association International Conference, confirm positive results announced in May.
The FDA recently approved the first cellular therapy for Type 1 diabetes and others may not be far behind. But experts say challenges still exist to the widespread application of these treatments.
Severe respiratory problems killed seven patients and affected five others, though the company said that most of these were likely unrelated to the drug.
After paying $300 million upfront to BeiGene for option rights to ociperlimab in December 2021, Novartis has dropped the agreement and given the rights back to the Chinese biotech.
Early-stage data shows that Viridian’s thyroid eye disease candidate induces clinically meaningful improvements in eye protrusion after six weeks of treatment.
Johnson & Johnson has licensed Nanobiotix’s lead radioenhancer candidate, designed to increase the efficacy of radiation treatment in cancers, capitalizing on the latter’s rocky financial situation.
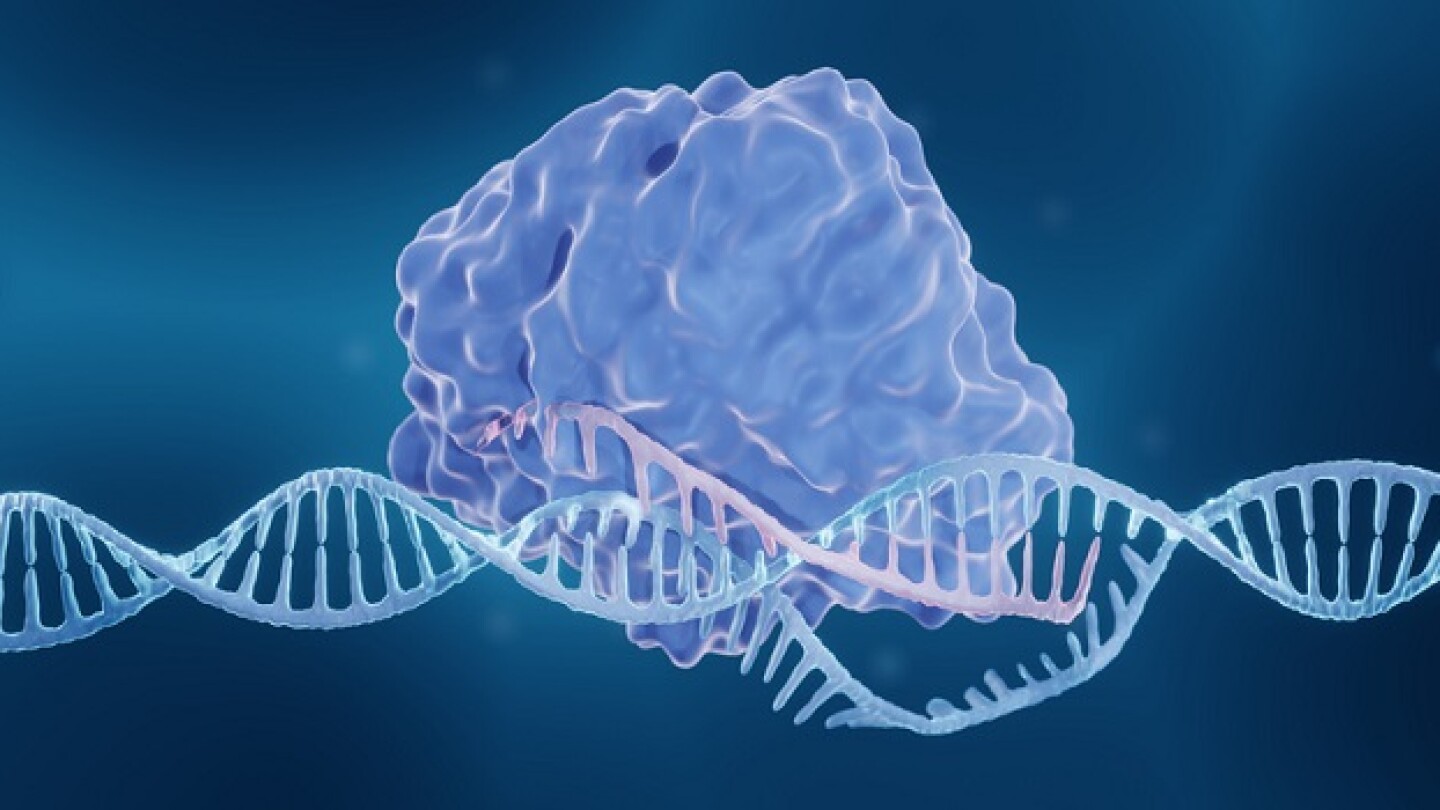
Gene editing technologies are advancing rapidly in the clinic, with the potential first approval later this year, but challenges remain.
After initial Phase II data for the oral IL-23 receptor antagonist licensed from Protagonist Therapeutics spooked investors, Janssen provided a fuller readout and advanced the candidate.
More than a month after failing its Friedrich’s ataxia trial, PTC Therapeutics’ vatiquinone has again missed its primary endpoint, this time in a mitochondrial disease-associated seizures study.
Phase I/II data on AbbVie and Genmab’s recently approved bi-specific antibody in a second cancer indication positions them to talk to regulators about filings to challenge Roche’s Lunsumio.
PRESS RELEASES