Europe
A new packaging and labeling manufacturing facility has derailed the potential approval of Malinckrodt’s investigational hepatorenal syndrome (HRS) treatment, terlipressin.
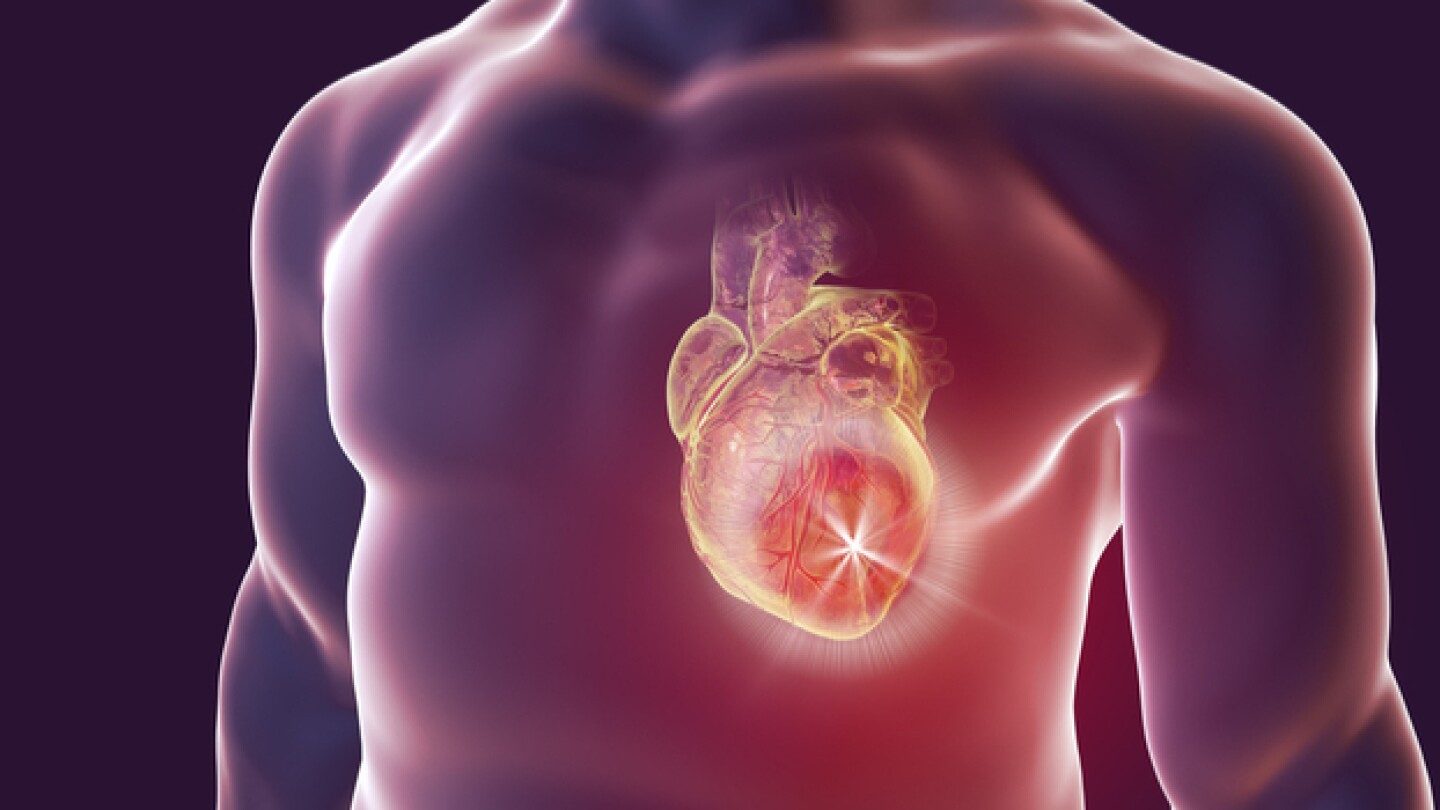
A new project uncovering genetic changes linked to damage following a heart attack could potentially pave the way for gene therapies to prevent long-term cell damage.
After roaring into February with positive data in five different indications, Regeneron and Sanofi announced today a late-stage Dupixent trial has been halted due to futility.
Bayer is heading back to the FDA to seek approval for a new indication of its prostate cancer drug Nubeqa, following positive Phase III results.
With the Omicron surge waning, at least in the U.S., there is speculation that the COVID-19 pandemic may be in its end stages. Some experts, however, warn that this could be premature.
Together, Intellia and ONK hope to develop, engineer and test new allogeneic CRISPR-edited NK cell therapies.
Roivant Sciences launches new subsidiary Hemavant that will develop Eisai-licensed asset for transfusion-dependent anemia in patients with low-risk MDS.
ProQR Therapeutics has reported that their drug sepofarsen failed to meet its primary and secondary endpoints in recent Phase II/III clinical trials.
Moderna is reportedly in talks with the UK government to establish mRNA manufacturing facilities in the country, as well as collaborating with health authorities on clinical trials.
The EMA’s safety committee has announced that they are reviewing reports of menstrual disorders after people received COVID-19 vaccinations from Pfizer-BioNTech and Moderna.
PRESS RELEASES