Europe
Hutchison China MediTech Limited (“Chi-Med”) shares additional analyses from three completed and ongoing clinical studies of fruquintinib and savolitinib at the 22nd Annual Meeting of the Chinese Society of Clinical Oncology (“CSCO”) on September 18 to 22, 2019 in Xiamen, China.
Summit Announces Publication of Editorial in Future Microbiology Advocating for Innovation in Antibiotic Development to Drive Stewardship Focus on Improving Patient Outcomes
Biom’up SA announced that Patrice Ferrand was appointed CEO, replacing Dr. Jan Ohrstrom. Dr. Ohrstrom, who was appointed CEO in May of this year, remains Chairman of the Board, a role in which he has served for the Company since 2015.
Dornier MedTech Launches New Image Post-Processing Software for Urology to Improve Kidney Stone Treatment Outcomes
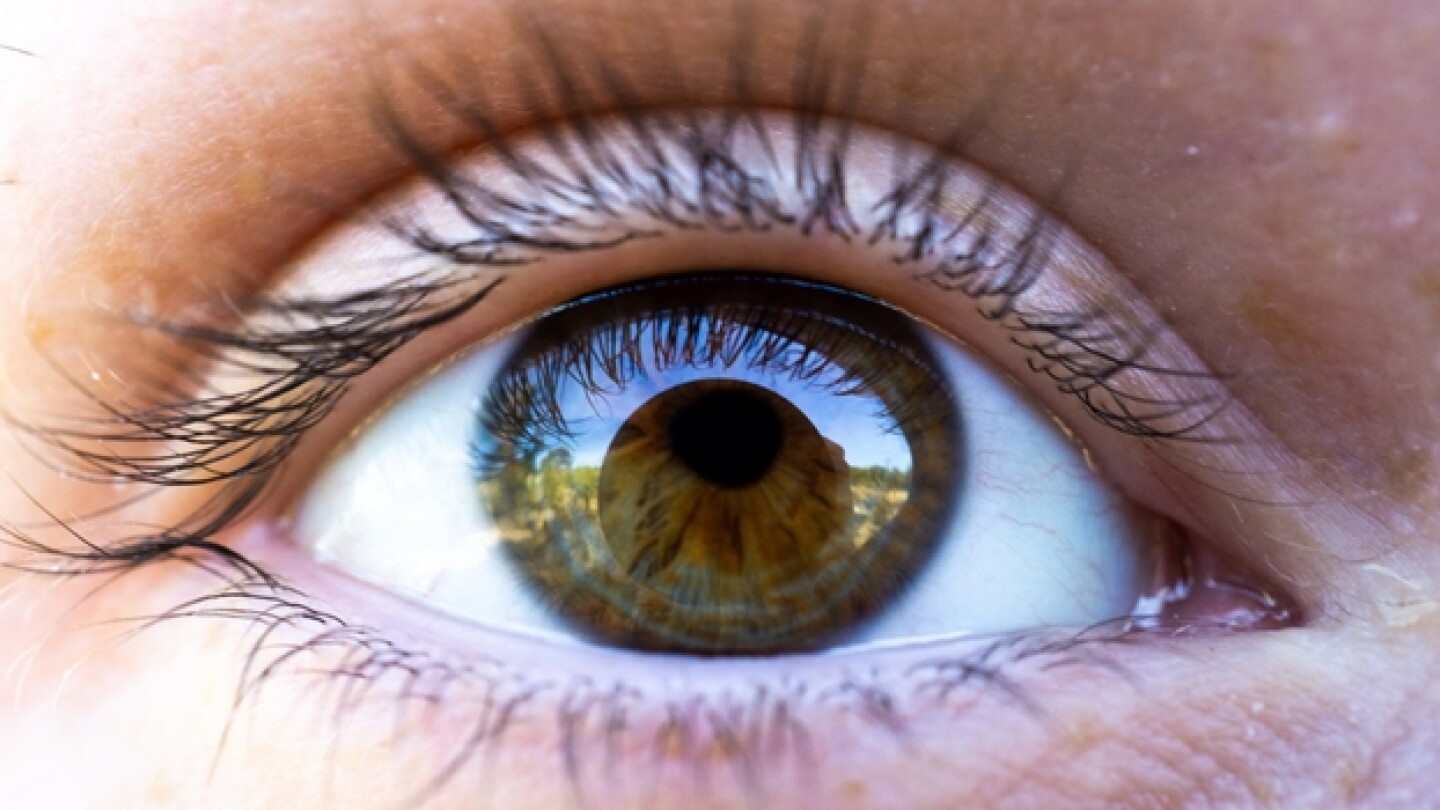
Researchers from Genentech and its parent company Roche have developed what is being called the first “deep learning model” that can predict which patients with diabetic retinopathy will progress the fastest.
RESCUE and REVERSE are two separate but parallel Phase III trials evaluating the efficacy of a single intravitreal injection of GS0101 in patients with LHON from the G11778A mutation in the mitochondrial ND4 gene.
Novo Nordisk’s oral-semaglutide for type 2 diabetes snagged approval from the U.S. Food and Drug Administration late Friday, marking it as the first such treatment to be approved in pill form.
Clovis Oncology plunked down $12 million in an upfront payment to Germany-based 3B Pharmaceuticals for licensing rights to peptide-targeted radionuclide therapy and imaging agent targeting fibroblast activation protein alpha.
Ipsen announced positive data from the ENGAGE Phase IIIb/IV clinical trial of Dysport (abobotulinumtoxinA) to treat upper and lower limb spasticity in adults with a Guided Self-rehabilitation Contract.
Last week was an unusually busy week for clinical trial news, with numerous companies presenting results at conferences. Here’s a look at the top stories.
PRESS RELEASES