Blade Therapeutics
NEWS
Closing out the month of May, plenty of companies presented or announced clinical trial updates and news. Here’s a look.
Heading into the middle of January, companies announced plenty of new clinical trial news. Here’s a look.
Going through the most newsworthy stories of the year, BioSpace found trends more than one big story, topics that just kept rising again and again. Here’s a look.
Heading out of November and into December, plenty of companies had clinical trial news to report. Here’s a look.
The merger between Blade Therapeutics and Biotech Acquisition Company will provide the company with $254.3 million to drive the development of cudetaxestat.
It was yet another busy week for clinical trial news. Here’s a look.
Please check out the biopharma industry coronavirus (COVID-19) stories that are trending for July 22, 2020.
Public perception of the industry can be a bit confusing. Life Science Leader recently pulled together several biopharma executives for a round table discussion of the biopharma industry’s public perception—generally bad—and what might be able to do about it.
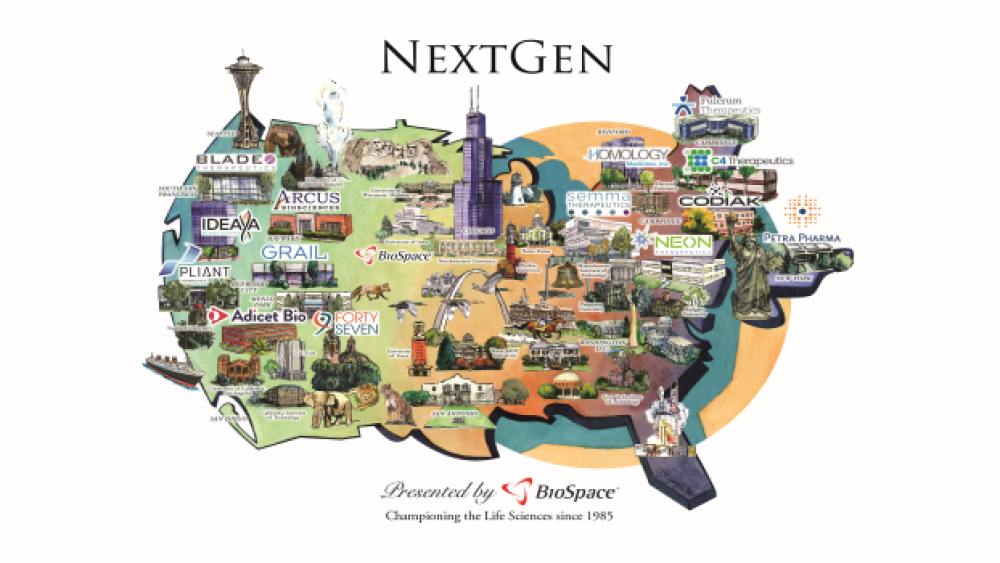
BioSpace is proud to present its NextGen “Class of 2017,” which is a list of 20 up-and-coming life science companies that launched no earlier than 2014.
JOBS
IN THE PRESS