AstraZeneca
NEWS
Has the U.S. Food and Drug Administration failed to do its due diligence in finding and reporting financial conflicts of interest between members of its scientific advisory committees and pharmaceutical companies?
Both “rare cancers” and “sub-populations of cancers” present a challenge to companies conducting clinical trials—finding enough patients for those trials.
Roche announced that its Phase III IMpassion130 clinical trial met its co-primary endpoint of progression-free survival (PFS) in triple-negative breast cancer (TNBC).
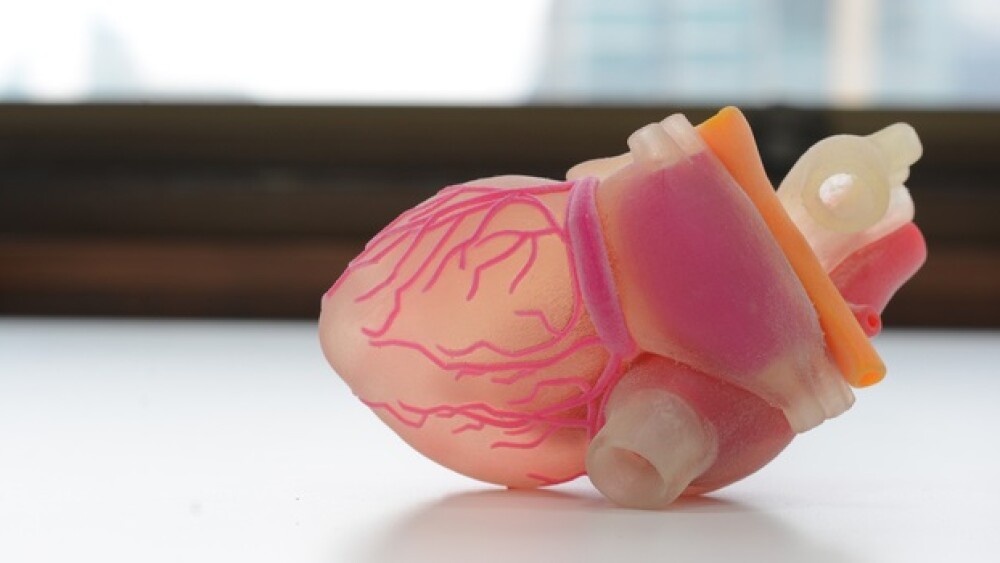
There has been recent coverage of 3D bioprinting and how it has the potential to create a brave new world of printed organs to be used in biopharma research and maybe someday to even be used in organ transplants.
This week had a few disappointing clinical trials, as do most weeks, but it also had a number of significant successes. Here’s a look at some of the clinical trials that met their primary endpoints this week.
AstraZeneca and Merck announced positive results from their Phase III SOLO-1 clinical trial of Lynparza (olaparib) in BRCA-mutated (BRCAm) advanced ovarian cancer.
As the European Medicines Agency prepares to make Amsterdam its new home following the U.K.’s Brexit, Holland is likely to see its biopharma industry snag a hoped-for boost in new business due to the presence of the regulatory agency.
Akero Therapeutics, headquartered in Cambridge, Massachusetts, closed on a $65 million Series A financing round.
Boston-based Emulate closed on a Series C round worth $36 million.
JOBS
IN THE PRESS