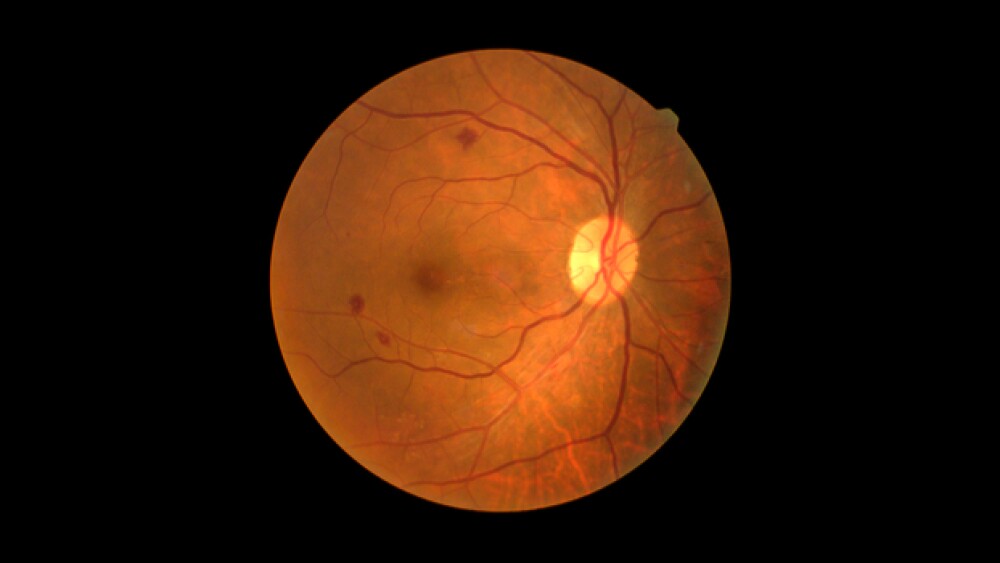
Aerpio Therapeutics
NEWS
Aerpio Therapeutics merged with Aadi Bioscience in 2021.
It was another busy week for clinical trial announcements. Here’s a look including trials for COVID-19, migraine, Parkinson’s disease, Alzheimer’s, HIV and more.
It was a busy week for clinical trial updates. Here’s a look.
Aerpio said the Phase IIb asset did show some positive data in a number of pre-specified secondary endpoints, including changes in Urine Albumin-Creatinine Ratio, a measure of kidney function, and in intraocular pressure.
Since it first launched with $100 million in the bank earlier this year, Gossamer Bio has been on something of a roll.
This morning two companies announced deals that will yield potential benefits to multiple patient populations.
JOBS
IN THE PRESS