Merck & Co.
AWARDS
NEWS
Dr. Anthony Fauci expects the FDA will authorize the Pfizer-BioNTech vaccine for children under the age of five in the next month.
A checkpoint inhibitor under development from China’s CStone Pharmaceuticals hit the mark in a Phase III non-small cell lung cancer study.
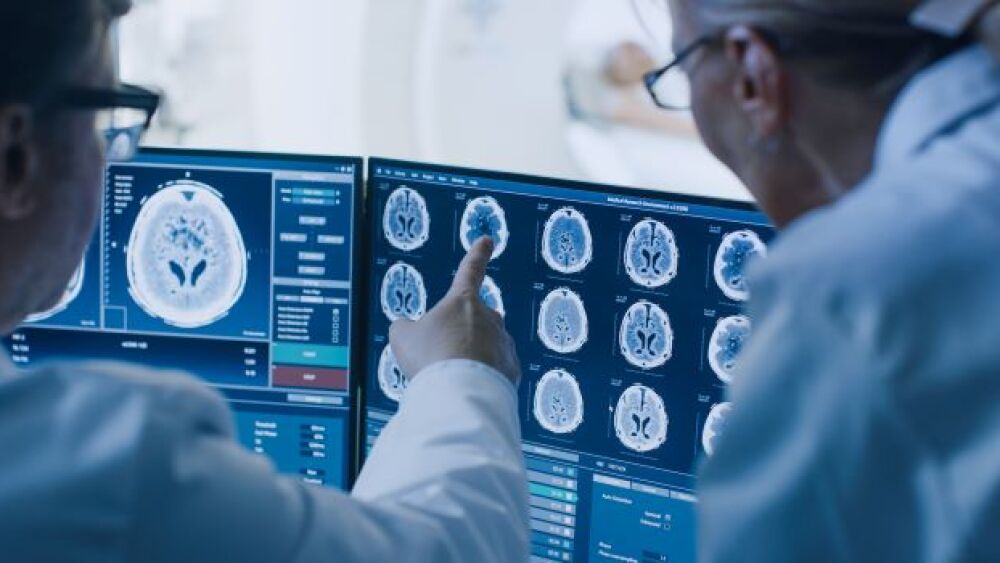
Aria Pharmaceuticals is using a unique computational approach to drug development that has increased its success rate by 30-fold when compared to traditional approaches.
Heading into the middle of January, companies announced plenty of new clinical trial news. Here’s a look.
Biopharma and life sciences companies strengthen their leadership teams and boards with these Movers & Shakers.
The approval was based on data from a broad clinical program of 1,854 adults with insomnia conducted at more than 160 clinical sites in 18 countries.
JOBS
IN THE PRESS