CRISPR Therapeutics
NEWS
Several biopharma companies are closing out the month of April with expansions to facilities, including a $3 billion investment to develop next-generation life science ecosystems.
Vertex has made a point over the past decade to make research and development a priority, investing over 70% of its expenses back into it.
The U.S. Patent and Trademark Office ruled that the use of CRISPR-Cas9 in humans belongs to Broad, not UC Berkeley.
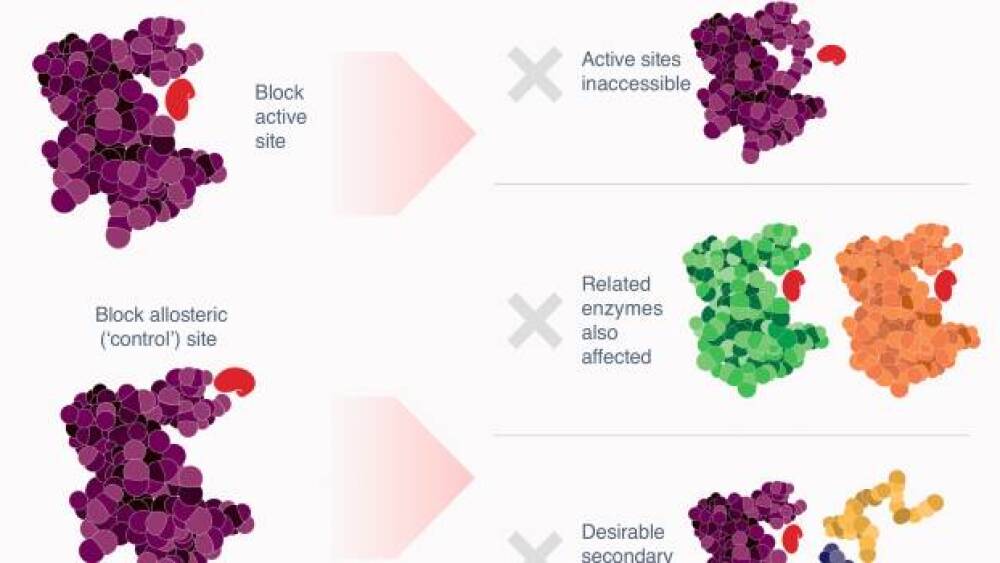
Exo Therapeutics’ approach is not only different, it has revolutionary potential.
It was a relatively quiet week for clinical trial announcements. Here’s a look.
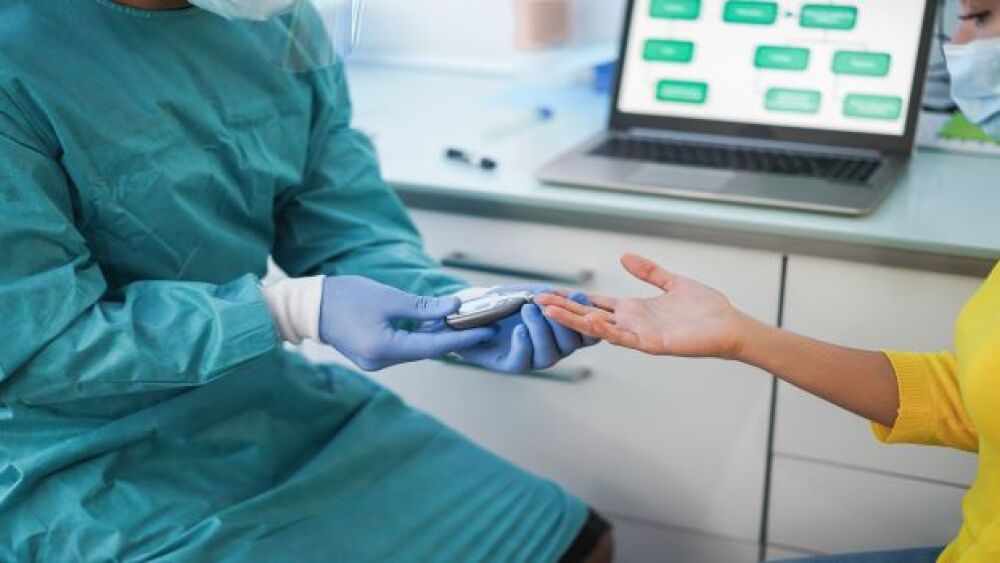
CRISPR Therapeutics and ViaCyte dosed their first patient in Phase I clinical trial of a novel gene-edited cell replacement therapy to treat Type 1 Diabetes.
Biopharma and life sciences companies from across the globe provide updates on their businesses and pipelines.
Sangamo Therapeutics announced that Sanofi US was returning its rights to SAR445136 as it shifts its approach from personalized cell therapies to allogeneic off-the-shelf genomic approaches.
Biopharma and life sciences companies from across the globe provide updates. Belgium-based Galapagos NV won European approval for Jyseleca for the treatment of adult patients with moderately to severely active ulcerative colitis (UC).
JOBS
IN THE PRESS