Spectrum Solutions Receives CE Mark Approval for its SDNA-2000 and SDNA-3000 Saliva Collection Devices used for IVD Molecular Diagnostic Testing
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210106005324/en/
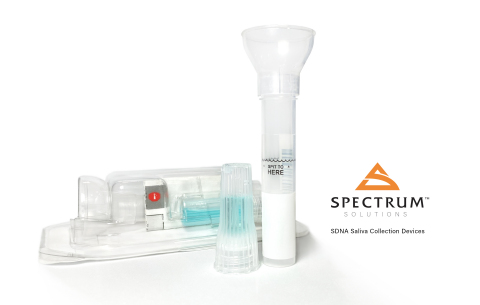
Spectrum Solutions receives CE Mark for SDNA Saliva Collection Device. (Photo: Business Wire)
“Receiving CE Mark approval for the SDNA-2000 and SDNA-3000 is a major milestone for our company and this offers physicians, healthcare organizations, governments, and individuals needing repeat IVD diagnostic testing an easy and pain-free option,” said Stephen Fanning, CEO of Spectrum Solutions. “At-home bio-sample self-collection means staying home when you don’t feel good and limiting exposure to other harmful illnesses for those most at risk.”
The Spectrum Solutions SDNA Saliva Collection Device was the first saliva-based diagnostic testing solution to receive the U.S. Food and Drug Administration (FDA) Emergency Use Authorization (EUA) for COVID-19 testing in the United States.
Bio-samples collected with the SDNA saliva collection system consistently demonstrate higher levels of testing accuracy, provide the safest & most robust testing biomaterial, is able to pinpoint an infection’s lifecycle stage, and delivers over 10 days of post-collection stability with no degradation in sample efficacy. The device’s intuitive ease-of-use design was engineered to facilitate unsupervised testing sample self-collection virtually eliminating user collection errors.
ABOUT SPECTRUM SOLUTIONS AND SPECTRUM DNA
Headquartered in Salt Lake City, Utah, Spectrum Solutions and its medical device and services division, Spectrum DNA, focus their concentrated industry expertise on engineering innovative end-to-end solutions for both clinical diagnostic projects and commercial product plans. Spectrum Solutions is a single-source provider of on-site medical device development and manufacturing, custom packaging, kitting, and direct-to-consumer fulfillment. Its bio-sample collection devices, patented technologies, and dedicated services deliver measurable process optimization, unprecedented efficiency, and unmatched global scalability. For more information, please visit spectrumsolution.com/SDNA.
View source version on businesswire.com: https://www.businesswire.com/news/home/20210106005324/en/
Source: Spectrum Solutions, LLC